The chemical building block of the atomic structure
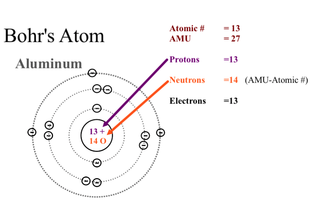
The Bohr model shows a simple model of the atomic structure.
The atom has basic structure with just a central core. A nucleus containing the protons(positive charged) and neutrons(neutral).The atom is the smallest part of an that can retain the properties of the element.
That atom can also be broken down in three subatomic particles that don't have the properties of that element.
- The first subatomic particle are positive charged protons.
- The second subatomic particle are neutral neutrons. Neutral meaning it is not positive nor negatively charged.
- And lastly, the third subatomic particle are negative charged electrons.
Review questions.
1. What type of charge are protons?
a. negative
b. neutral
c. positive
d. battery
2. Explain what the Bohr helps do.
3. The structure of an atom is complex.
a. true
b. false
4. Draw a Bohr model without looking at one for reference.
5. How many subatomic particles are there?
a. 7
b .5
c. 1
d. 3
a. negative
b. neutral
c. positive
d. battery
2. Explain what the Bohr helps do.
3. The structure of an atom is complex.
a. true
b. false
4. Draw a Bohr model without looking at one for reference.
5. How many subatomic particles are there?
a. 7
b .5
c. 1
d. 3
